Binding assays (binding affinity assays – radioimmunoassays) are mainly run to confirm conservation of binding properties of radiolabeled compounds before ADME/DMPK investigations (even if chelation or iodination induces minor modifications of biologics, it is essential to demonstrate that they retain their biological properties). Binding assays can also be designed for specific kinetic binding experiments.
Internalization assays and cytotoxicity assays are conducted during the development of radiopharmaceuticals. These cell-based assays help to better understand mechanisms of therapeutic radiolabeled antibodies (or radiolabeled peptides) whose mode of action can require internalization.
Binding Assay
Our team uses both direct and competitive binding assays. A binding assay measures the interaction of a radiolabeled compound to its specific antigen. This affinity is a key characteristic of drug candidates under development.
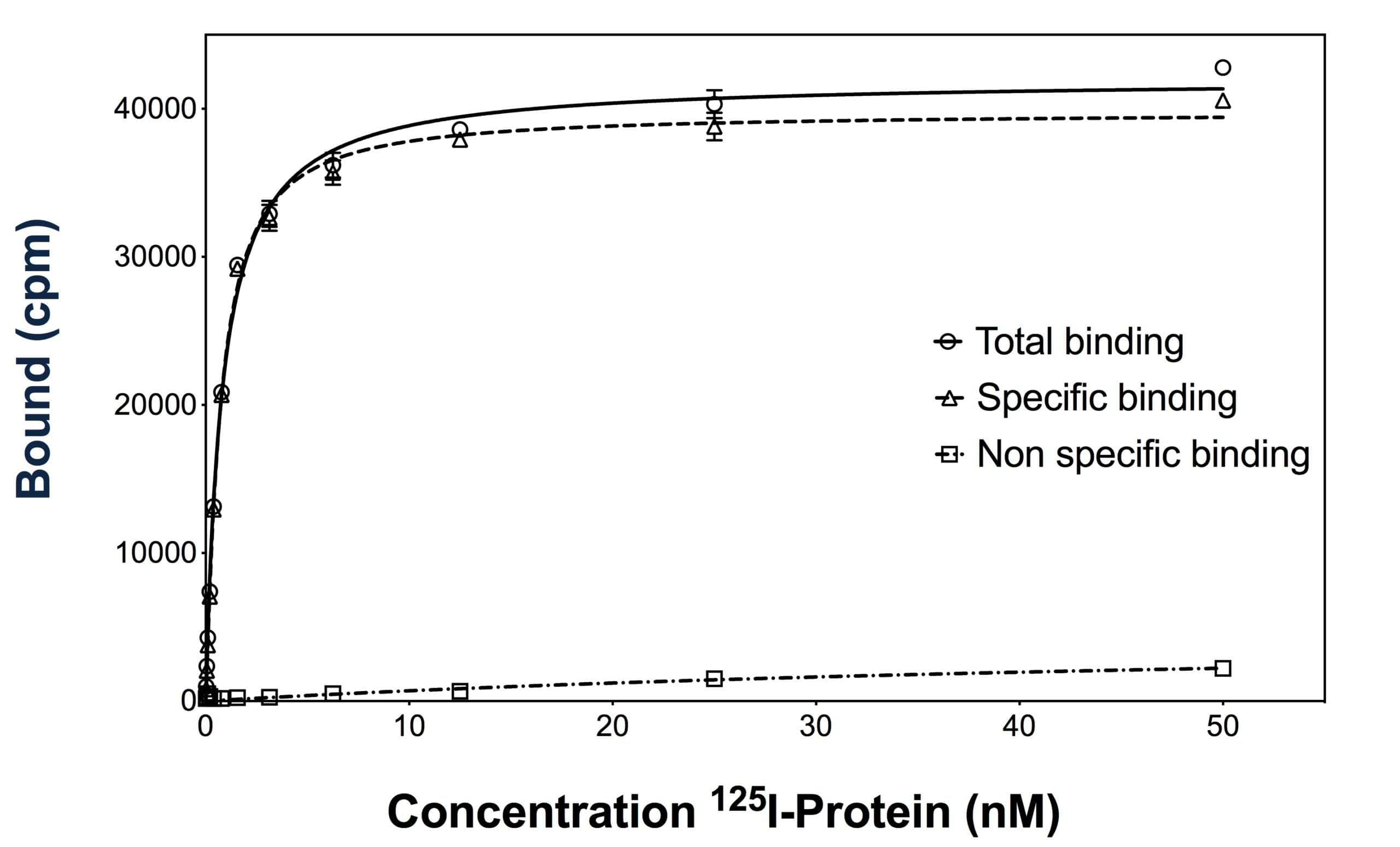
- Radioactive binding assays will allow to determine the dissociation constant (Kd) of the radiolabeled compound and the receptor density (Bmax) when running direct binding assays
- Competitive binding assays will allow to determine IC50 & inhibition constant (Ki) of competitive molecules
- Kinetic binding experiments can also be proposed in order to define association and dissociation rate constants (kon and koff)
Cytotoxicity Assay
Among all cell-based assays, cytotoxic assays evaluate the toxic potential of therapeutics designed to deliver specific payloads to cells (of either radiolabeled compounds or ADCs).
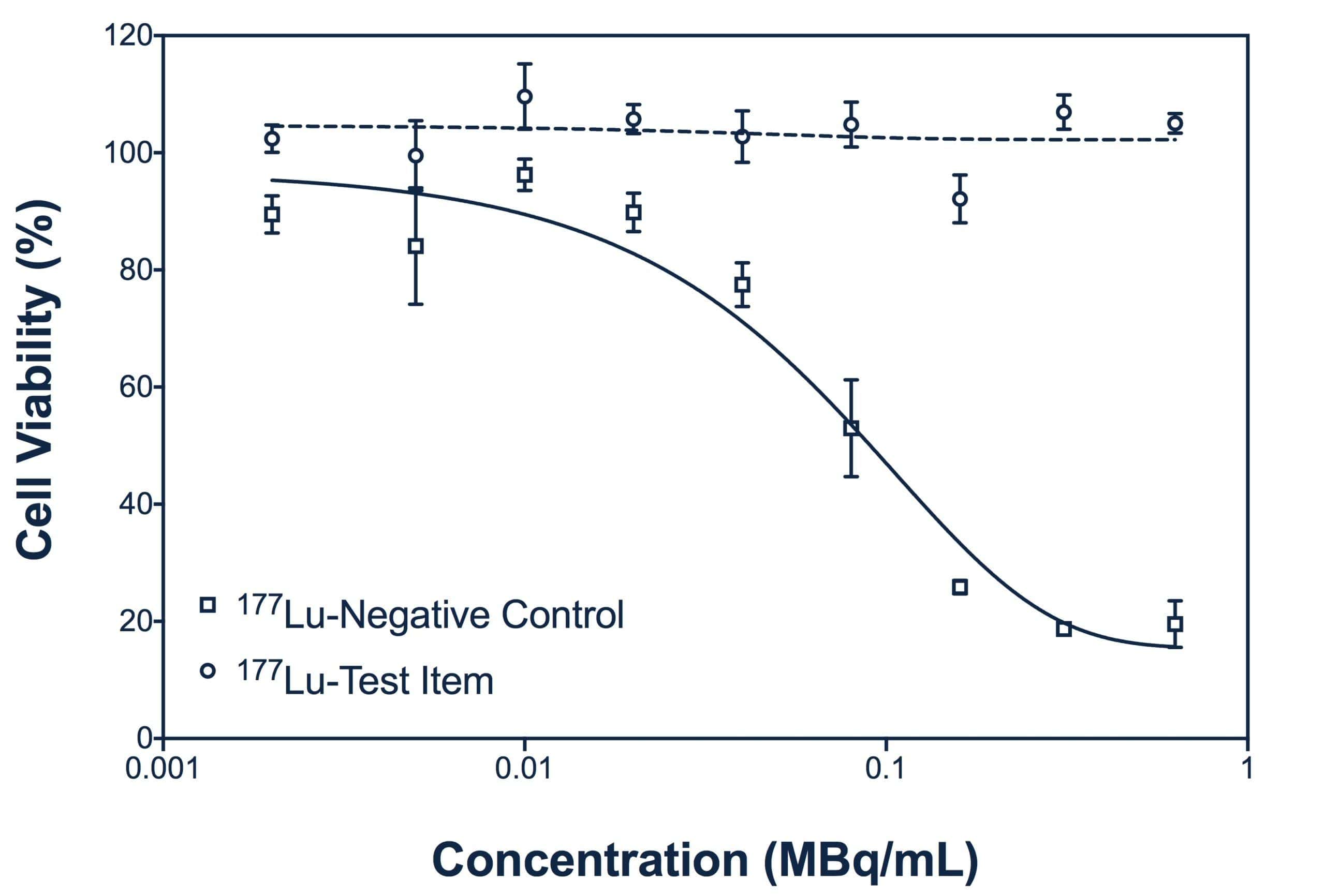
Cell viability will be evaluated using specific indicators of the cellular metabolic activity such as MTT or MTS. After optimization, a cytotoxic assay provides an efficient, rapid and reliable test to compare several candidates.
Internalization Assay
Internalization assays (specific cell-based assays) evaluate the therapeutic potential of a radiolabeled drug that not only binds to antigens expressed on tumor cells membranes but also are internalized.
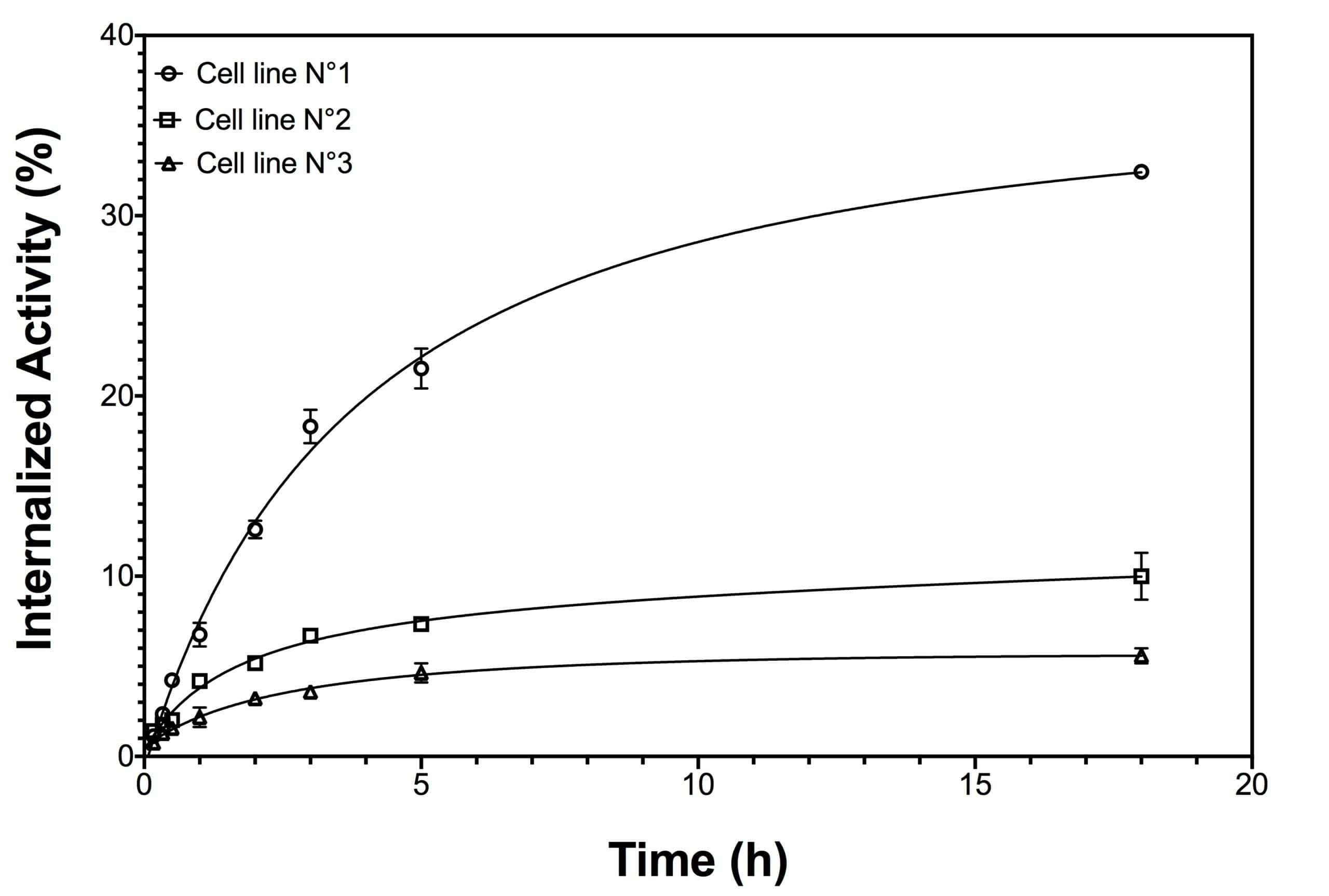
This cell-based assay quantifies:
- the kinetics of internalization
- the internalization rate
It distinguishes radiolabeled compounds simply associated with cell surface receptors as opposed to taken up fraction.
For more than 20 years, Chelatec provides in vitro services using radiolabeling technology. Our highly-experienced team develops tailored cell-based assays and any binding assays using soluble receptor to support preclinical drug target validation.
Chelatec will accurately provide you with the critical data you need to validate your drug candidate and can further assist you in conducting in vivo investigations in relevant mouse models.
